Innovations
Thai Otsuka Pharmaceutical Co., Ltd, has realized the importance of products that affect the efficiency of medical treatment. Therefore, we have performed a dedicated research and development of Pharmaceutical Products and Medical Devices to be imported for the patients in Thailand.
In addition to the quality of the products, the quality of personnel is also important that must be simultaneously developed to ensure that the personnel of the Company can effectively communicate the key scientific messages to Thai Healthcare Professionals.
Since Thai Otsuka Pharmaceutical Co., Ltd. has recognized the importance of the Thai medical society, we have been involved in promoting the research and development of knowledge in the treatment of patients for more efficiency and pushing the establishment of various medical associations in Thailand.
Therefore, all employees of Thai Otsuka Pharmaceutical Co., Ltd. are extremely proud to be a part of an organization that has a strong sense of social responsibility and that stands by Healthcare Professionals to develop the treatment and the health promotion of Thai people.


GMP-BSI
Beverages in sealed containers, foods in sealed containers (Food with low water activity) (Special purposed foods) was manufactured in Thai Otsuka Pharmaceutical Co., Ltd. whose GMP 193 standard is certified by BSI Group (Thailand) Co., Ltd. 002/2557 Registered by Thai FDA.

PICs GMP
Terminally sterilised Products (Large volume liquids and Small volume liquids) was manufactured in Thai Otsuka Pharmaceutical Co., Ltd. whose PICs GMP is certified by Food and Drug Administration, Ministry of Public Health, Thailand.

ISO 9001
Sterile Drug Products (Small Volume and Large Volume Sterile Solution) and Enteral Nutrition Products was manufactured in Thai Otsuka Pharmaceutical Co., Ltd. whose ISO 9001 standard is certified by SGS (Thailand) Ltd.

ISO 14001
Sterile Drug Products (Small Volume and Large Volume Sterile Solution) and Enteral Nutrition Products was manufactured in Thai Otsuka Pharmaceutical Co., Ltd. whose ISO 14001 standard is certified by SGS (Thailand) Ltd.

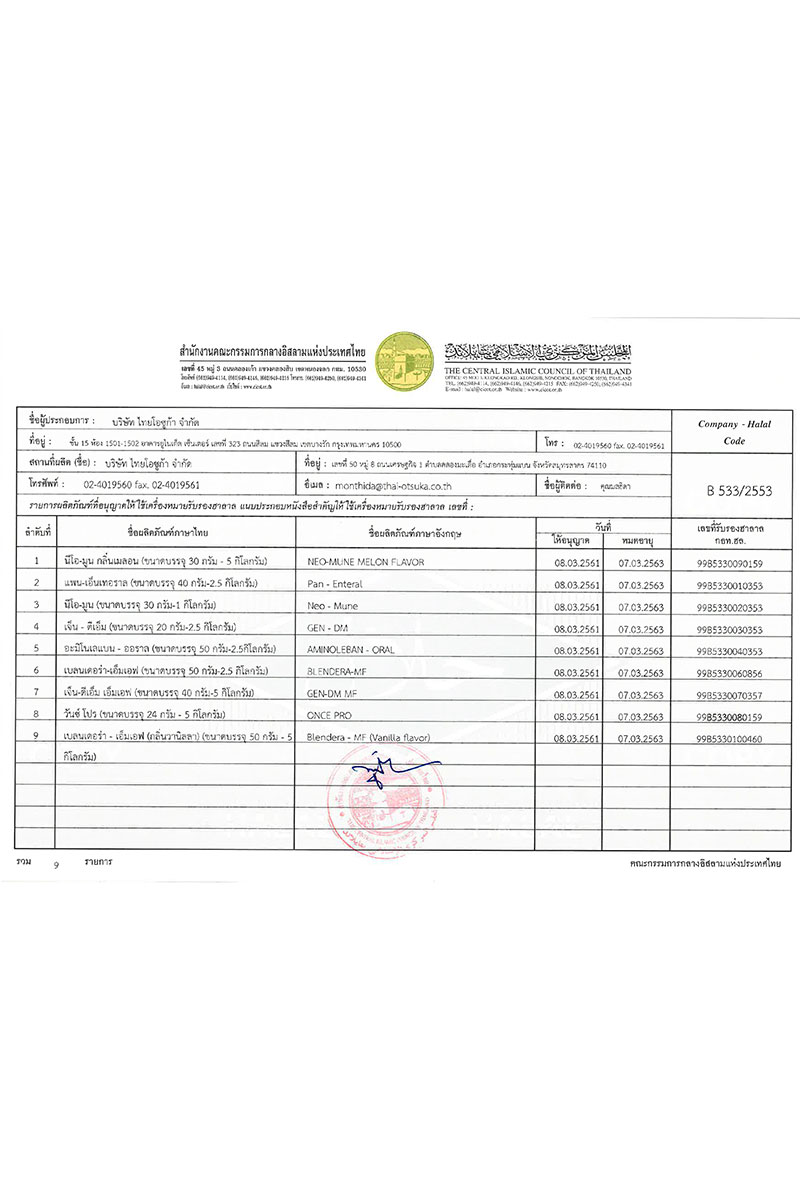
HALAL
Neo-Mune (Melon flavor), Pan-Enteral, Neo-Mune (Vanilla flavor), Gen-DM, Aminoleban-Oral, Blendera-MF,Gen-DM MF, Once Pro and Blendera-MF (Vanilla flavor) was manufactured in Thai Otsuka Pharmaceutical Co., Ltd. whose HALAL is certified by The Central Islamic Council of Thailand.

Committed to continuous research and development of medical food products.
To create a better quality of life for our consumers
Thai Otsuka Co., Ltd. has established a research and development unit with the purpose to research and develop new medical food products to respond to the diverse needs of health consumers whether it be a specialized medical food or medical food for general health care providers.
The study of consumer insights from marketing research, reaching the needs of the consumer as much as possible ,clinical research with medical staff from the hospital including leading institutions in the year 2017, Thai Otsuka Co., Ltd. joined the Thai Government’s Food Innopolis project to develop and research medical food and progress with more advanced knowledge and technology.



Invent, develop and
standardize production.
We are also passionately building a company devoted to better health.
Our products have been internationally recognized
and can be exported to many countries and regions such as Japan, Taiwan,
Hong Kong, Indonesia, Philippines, Vietnam, Malaysia,
Pakistan, Myanmar and Cambodia.
These countries and regions have high standards
for the selection of imported product,
especially Japan.